
Can LiMnO2 Batteries Be Charged: Lab Test Results & Industrial Solutions
If an engineer asks, “can LiMnO2 batteries be charged,” the risk is already present: a wrong charger, a reused holder, or a field technician assuming all coin or cylindrical lithium cells behave like Li-ion cells. Misuse can cause leakage, voltage collapse, or unsafe heat.
The answer is no. LiMnO2 battery charging is not an approved operating mode because lithium manganese dioxide chemistry[1] is designed as a primary system, not a reversible storage system.
A non-rechargeable LiMnO2 cell can deliver stable energy for industrial devices, but forced charging may trigger gas generation, lithium plating, seal damage, leakage, and permanent loss of safety margin.
The better question is not only can LiMnO2 batteries be charged, but how to design products so nobody tries.
Table of Contents
- 1. Why LiMnO2 Cannot Be Recharged?
- 2. What Happens If You Try to Recharge?
- 3. Which Engineering Solutions Prevent Misuse and Support Safer Battery Design?
1. Why LiMnO2 Cannot Be Recharged?
LiMnO2 cells cannot be safely recharged because their electrochemical reaction is not engineered for repeated lithium insertion and extraction[2]. In lithium manganese dioxide primary cells, discharge changes the cathode structure and internal chemistry in ways that are not reliably reversible.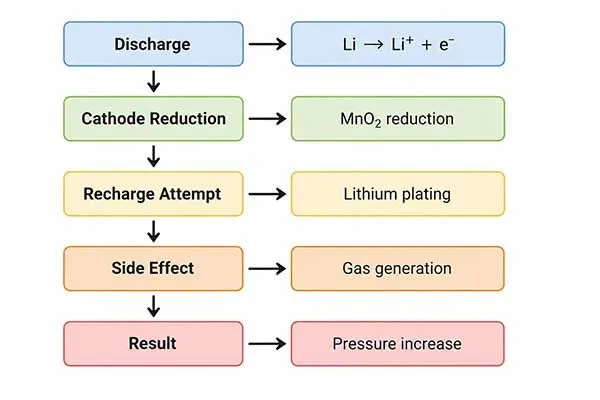
Therefore, “can LiMnO2 batteries be charged” should be answered as no for normal industrial system design.
Chemistry limits, engineering selection, and system design
In engineering selection, the key distinction is primary function[3] versus cycle function.
A LiMnO2 cell is optimized for stored energy, shelf life, low leakage current, and predictable discharge under a defined load. It is not optimized for accepting external current.
In lithium manganese dioxide product reviews, the first gate question is usually “can LiMnO2 batteries be charged”, because the answer decides connector, labeling, and protection design.
During discharge, lithium ions move and the manganese dioxide cathode changes state. After meaningful discharge, returning the cell to its original structure is not a controlled, repeatable process.
This is why non-rechargeable LiMnO2 cells appear in memory backup, safety equipment, metering modules, medical accessories, alarms, trackers, and industrial electronics where the product sleeps for long periods and then wakes up reliably.
| Design factor | LiMnO2 primary cell behavior | Engineering implication |
|---|---|---|
| Electrode reversibility | Not designed for repeated cycling | Do not specify a charge path |
| Normal use case | Long standby and controlled discharge | Good for low-maintenance industrial products |
| Misuse mode | External current may create internal side reactions | Use electrical and mechanical anti-charge controls |
| Keyword distinction | LiMnO2 battery charging is not equivalent to Li-ion charging | Separate BOM, labeling, and service process |
However, in system design, the charger interface must be excluded. If a PCB shares a port with a rechargeable platform, the firmware, connector, protection circuit, labeling, and service manual should all assume that someone may ask can LiMnO2 batteries be charged and may act incorrectly unless the design blocks it.
The comparison with LiMnO2 vs Li-ion battery is often misunderstood. Li-ion cells use charge management, protection ICs, controlled voltage limits, and cycle-rated electrodes. LiMnO2 cells are selected for primary duty, not cycle recovery.
2. What Happens If You Try to Recharge?
If you try to recharge a LiMnO2 cell, the cell may not simply “store energy again”. It may heat up, generate gas[4], leak electrolyte, swell, lose capacity, or fail under later pulse load. Don’t charge the Li-MnO2, especially for safety, healthcare, metering, and high-reliability backup power systems.
Real Lab Test
In a controlled lab context, charging a discharged LiMnO2 sample above its intended voltage window may initially look harmless because the voltage rises. That observation is misleading.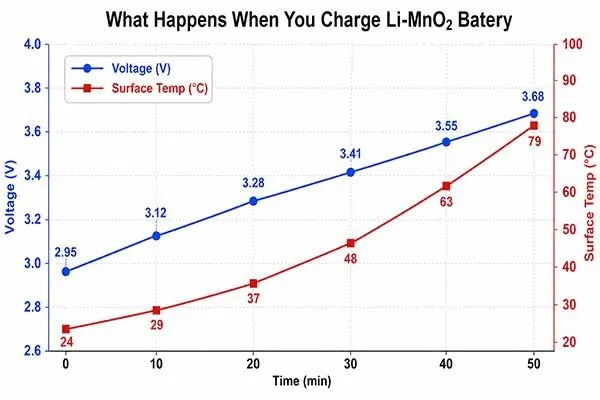
Voltage recovery does not prove chemical reversibility. When external current is forced into a primary cell, side reactions can consume active material, create gas, damage the seal, or increase internal resistance. The cell may pass a short open-circuit voltage check and still fail a pulse-load test later.
From a manufacturer’s view, the field risk is not only one abused cell. The larger risk is a system-level habit: technicians using a universal bench charger, a maintenance team reusing a Li-ion dock, or a product designer leaving a USB charging path connected to a primary battery rail.
| Observed condition | Possible internal mechanism | System-level risk |
|---|---|---|
| Voltage rises during forced charge | Surface polarization, not reliable recovery | False confidence during incoming inspection |
| Temperature increases | Side reactions and internal resistance growth | Thermal stress near plastic housings |
| Leakage after overvoltage | Gas pressure and seal damage | Corrosion, contamination, customer complaint |
| Poor pulse response | Higher impedance after abuse | Boot failure, alarm failure, or data loss |
This is why primary lithium battery charging[5] must be treated as a misuse condition in the risk analysis, not as a backup feature.
Lab evidence should be interpreted conservatively: even if a cell accepts some current, that does not make it rechargeable, certified, or safe for customer equipment.
3. Which Engineering Solutions Prevent Misuse and Support Safer Battery Design?
Engineering solutions include eliminating charge paths, adding blocking diodes[6] or ideal-diode circuits[7], using PTC devices, fuses, TVS protection, low-voltage cutoff, clear non-charge labeling, and selecting secondary chemistry when true recharging is required.
For can LiMnO2 batteries be charged, the safe design answer is to prevent charging and choose alternatives when cycling is required.
Practical protection architecture and custom battery evidence
System design should begin with a misuse map, because can LiMnO2 batteries be charged is a question that often appears only after a product already has a shared power port.
Ask where current could enter the battery: USB ports, docking pins, test pads, solar inputs, reverse-installed packs, service adapters, or external power rails. Then block those paths physically and electrically.
A simple diode may be enough for low-current equipment, while higher-efficiency systems may need an ideal-diode controller. For surge environments, a TVS diode, resettable fuse, polarity protection, and low-voltage cutoff can reduce abuse severity.
If the product must be charged repeatedly, do not debate can LiMnO2 batteries be charged; change the chemistry to a true secondary system after reviewing primary vs secondary lithium requirements.
A real customer case illustrates the point. A Japanese industrial portable ultrasound equipment manufacturer originally used two 18650 cells in series. The project required European medical certification standards, 5-year maintenance-free operation, less than 10% capacity loss at 25°C over 5 years, and instant high-pulse support.
As long life lithium primary battery manufacturer, we recommended a CR17505-based 12V 7000mAh pack with 2A maximum continuous discharge, 4A pulse discharge for 5 seconds, 45x45x55mm size, and 210g weight. The pack included a PTC thermistor, reverse-connection diode[8], and low-voltage cutoff circuit for non-chargeable protection.
During testing, the motor and amplifier pulled 4.5A at startup, battery-pack voltage dropped to 6V, and the system rebooted. The solution was to add an HPC1520 capacitor in parallel, recommend a revised startup sequence, and add a soft-start circuit.
Later, a user attempted charging above 15V, causing leakage. We added reverse-connection and overvoltage protection using a TVS diode plus resettable fuse, and laser-engraved “Do Not Recharge” on the housing.
| Engineering decision | Recommended action | Reason |
|---|---|---|
| Need long shelf life | Use non-rechargeable LiMnO2 or LiSOCl2 after load review | Primary cells support long standby better than many secondary packs |
| Need repeated charging | Consider Li-ion, NiMH, or rechargeable cr2032 where appropriate | The answer to can LiMnO2 batteries be charged is still no |
| Need manganese chemistry with cycling | Evaluate certified rechargeable batteries manganese options carefully | Do not confuse marketing names with primary cell behavior |
| Need supplier support | Work with Li-MnO2 lithium battery suppliers that test pulse, leakage, insulation, and protection behavior | Battery-pack reliability depends on the cell, circuit, and enclosure together |
For industrial purchasing, OEM lithium primary battery supplier support should include cell matching, PCM or protection-circuit design, connector selection, pulse-load verification, aging checks, insulation testing, vibration review, shipment documentation, and application-specific drawings.
Cost matters, but preventing recharge misuse is usually cheaper than handling leakage, returns, or field redesigns.
Conclusion
LiMnO2 cells are valuable primary batteries, but they should not be recharged. For engineers asking can LiMnO2 batteries be charged, the correct decision is chemical, electrical, and procedural: block charging, label clearly, validate pulse loads, and select a secondary chemistry when cycling is required.
A strong battery design review compares LiMnO2 vs Li-ion battery options, confirms primary-cell limits, and turns misuse scenarios into protection circuits before the product reaches customers. That is practical reliability engineering, not conservative wording for compliance.
Frequent Asked Questions about LiMnO2 Battery Charging
(Click to Unfold)
Q:How to recharge a button cell battery?
A:Most Li-MnO2 button cell batteries are primary batteries and are not designed for recharging. Attempting to recharge them may cause lithium plating, gas generation, leakage, or safety failure. For rechargeable applications, engineers should select lithium-ion coin cells or secondary lithium battery systems instead.
Q:What function does manganese dioxide have in batteries?
A:Manganese dioxide (MnO2) acts as the cathode material in Li-MnO2 batteries. It supports the electrochemical reduction reaction during discharge, helping deliver stable voltage, high energy density, low self-discharge, and reliable performance in industrial and backup power applications.
Q:Is the Li-MnO2 battery rechargeable?
A:Standard Li-MnO2 batteries are generally non-rechargeable primary lithium batteries. They are optimized for long shelf life and stable discharge rather than cycling performance. Recharging may damage the internal structure and create safety risks, especially in sealed cylindrical or coin cell designs.
Q:How to wake up a dead lithium battery?
A:A deeply discharged lithium battery may sometimes recover using a compatible charger with low-current pre-charge protection. However, primary Li-MnO2 batteries should not be reactivated or recharged after depletion. Industrial systems should replace exhausted cells to maintain safety and reliability.
Q:What is the voltage of Li-MnO2 battery?
A:A Li-MnO2 battery typically has a nominal voltage of 3.0V. Depending on the cell design and discharge conditions, the open-circuit voltage may range from approximately 3.0V to 3.3V, providing stable power output for medical, metering, IoT, and security devices.
Note:
[1]Learn how LiMnO2 fits into primary lithium battery families.↪
[2]See how lithium-ion movement differs between discharge and charge.↪
[3]Review primary battery chemistry choices for long-life devices.↪
[4]Review manufacturer warnings on gas, leakage, swelling, and fire risk.↪
[5]Confirm why primary lithium cells must not be treated as rechargeable.↪
[6]Learn how diode-based protection blocks reverse current paths.↪
[7]Compare ideal-diode controllers for low-loss reverse current protection.↪
[8]Compare reverse-battery protection methods for safer field service.↪



